Pharmaceutical & Life Science Projects
For over three decades, Simons Design has been building excellent working relationships within the pharmaceutical and Life Sciences Sectors, working with leading names such as GSK, Astra Zeneca, Actavis and Abbot Laboratories, as well as many start-up research companies and science park providers. Simons Design is a Technical Professional Service (TPS) Providers and IPD (Integrated Project Delivery) Partner to GSK providing Architectural Design Services to GSK across their UK sites.
Related Projects

U2 Building
The U2 building created a facility to support the respiratory product stream manufacturing of asthmatic inhalers for the US market. The building forms phase 1 of a major new development and production facility. A fast-track project, taking one year from inception to completion and product manufacture, the building was designed to enable fit-out to progress in tandem with the external fabric and to allow work to progress at all levels.
Client: GSK
Involvement: RIBA stages 0-7
Construction Value: £12million
Ground Floor: 1200 sqm
Mezzanine: 350 sqm
First Floor: 1,000 sqm

U3 Building
The U3 building was extended to increase manufacturing capacity for the Diskus device and support new inhaler technology. Delivered within a tight 12-month programme, the design enabled simultaneous external and internal construction.
The three-storey extension includes production, assembly, and packing areas, along with offices and support spaces. Built to FM standards, it features concrete foundations, a steel frame, and hollow rib floors. An innovative structure allows a column-free ground floor by suspending the first floor from above, creating open manufacturing space. The building is also connected to the existing respiratory facility to share support services.
Client: GSK
Involvement: RIBA stages 0-7
Construction Value: £17.5 million
Ground Floor: 1650sqm
First Floor: 1650sqm
Second Floor: 1650sqm

Completed: June 2014
Start on Site: January 2014
Handover: December 2015
M2 Building
New Build 3 storey OSD Manufacturing Facility, the facility maximised off-site and modular construction to minimise activities on an otherwise constrained site. The second floor plant room was fabricated and fitted off-site in 39 modules. Simons Design provided Architectural Design and Structural and Civil Engineering Designer services to develop the Scheme. Detail Design and Construction information, and provide technical support throughout the construction phase. Simons Design were appointed as an IPD Partner as part of an integrated project Delivery procurement strategy.
Client: GSK
Involvement: RIBA stages 0-7
Construction Value: £17.5 million
Ground Floor: 2500sqm
First Floor: 1350 sqm
Mezzanine Floors: 350 sqm
Second Floor: 1225 sqm
Scheme Design Start: August 2020
Design Completed: September 2021
Start on Site: October 2021
Handover: December 2023

Human Performance Centre
Project involved the remodelling of ground floor areas to form a new movement lab and briefing room. The first floor was remodelled to form new sensory labs, product branding lab and support areas.
Simons Design provided architectural design services to develop Concept, Scheme and Detail Design proposals and provided technical support throughout the pre-construction and construction phases, to handover.
Client: GSK
Construction Value: £2 million
Involvement: RIBA stages 0-7
Concept Design Commenced: February 2016
Start on Site: Sept 2016
Handover: June 2017

LAS Research Facility
This project involved the refurbishment of the existing LAS Facility at the GSK Stevenage site. This featured the facilitation of the relocation of the transgenics facility, associated laboratory and ancillary accommodation. This also included changing rooms, VHP chambers, storerooms and office areas. In addition to these works, the project also included the refurbishment of the building rack and tunnel washing facility and the associated ancillary accommodation.
Client: GSK
Construction Value: £7 million
Involvement: RIBA stages 0-7
Commissioned: March 2009
Start on Site: October 2009
Completion on site: March 2010
Operational: July 2010

Oral Solid Dose Pilot Plant Building
This project involved the remodelling of Flexible Clean Room Facilities and a storey extension.
The remodelling works included the development of a new mezzanine floor to support services distribution and maintenance outside of the cleanroom environments.
The facilities have incorporated optimum flexibility in that they allow expansion of suites both in plan and height to accommodate new processes and equipment in the future.
Client: GSK
Construction Value: £10 million
Involvement: RIBA stages 0-7
Ground Floor: 1500sqm
Mezzanine Floor: 1200sqm
Concept Design Commenced: February 2015
Start on Site: January 2017
Completed: December 2017
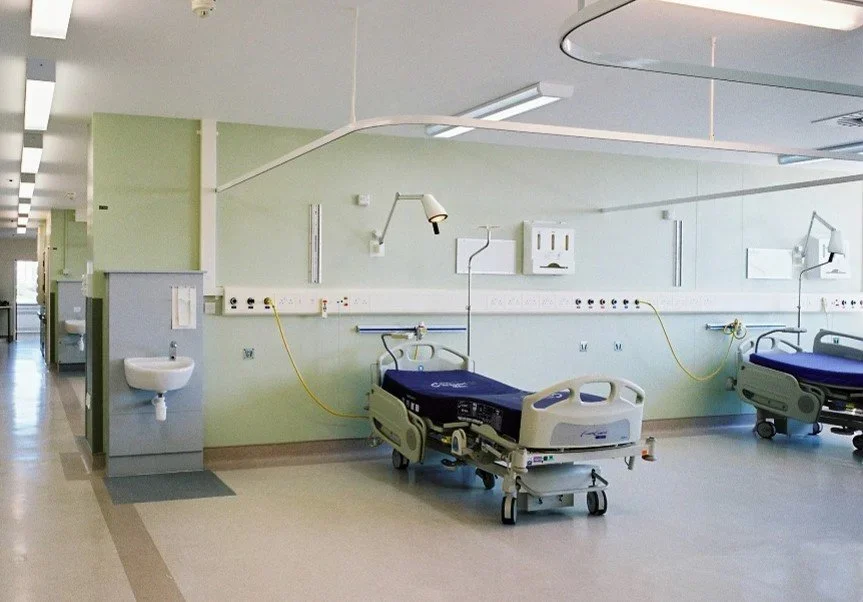
High Dependency Unit, QMC
Refurbishment of existing ancillary ward accommodation to create two four-bed high dependency wards and remodel the existing patient and support facility, improving monitoring of patients and staff facilities. Minimise disturbance to existing fabric due to location on 4th floor and occupied facility adjacent, above and below. Improve visitor and family waiting areas and the staff breakout facilities.
Remodel existing six-bed wards to four-bed wards. Creation of a new nurses’ station located to improve patient monitoring. Remodel exist single bed facilities and create new en-suites. Improved patient care through better monitoring and observation of patients through the improved design.
Client: Nottingham NHS Hospitals and Queen’s Medical Centre.
Construction Value: £350K
Involvement: RIBA stages 0-7
8 week construction phase

Drug Testing Facilities
Drug Testing Facilities for the London 2012 Olympic and Paralympics Games. The project involved the remodelling of existing Laboratory Facilities to accommodate both Olympic and non Olympic Drug testing facilities for sample testing prior to the commencement of the Games and the Games Period. The facility included analytical Laboratories, Blood and EPO Testing Laboratories and Ancillary Sample Storage and Staff Welfare Facilities.
Client: GSK & London Olympics
Construction Value: £1.8million
Involvement: RIBA stages 0-7
Ground Floor: 2750sqm
First Floor: 500sqm
Detail Design Completed: December 2010
Start on Site: January 2011
Handover: July 2011







